1.09 مليون برميل يوميا إجمالي إنتاج النفط الخام والمكثفات النفطية فـي سلطنة عمان أغسطس الماضي - جريدة الوطن

لغة الأرقام on X: "دليل حساب إجمالي دخل النفط في الموازنة من شرح دليل موازنة 2015 نلاحظ ان الحكومة تتحمل كلفة 40.8% من اجمالي الإنتاج و الشركات تتحمل تكلفة 59.2% تكلفة إنتاج

شاهد ملخص انتاج سلطنة عمان من النفط والغاز لشهرمارس وكم سعر بيع برميل النفط التقرير يبشر بخيركبير - YouTube
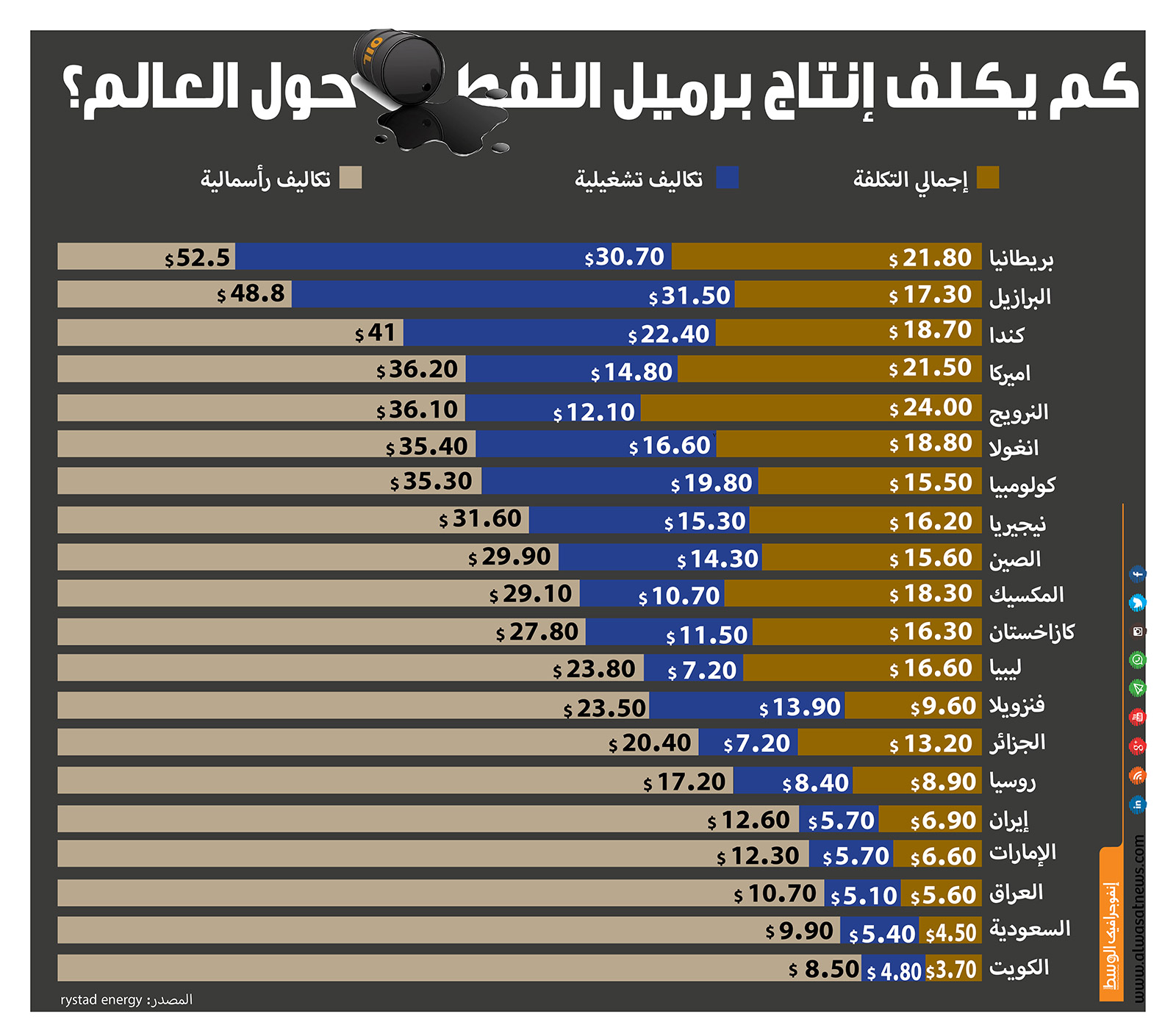
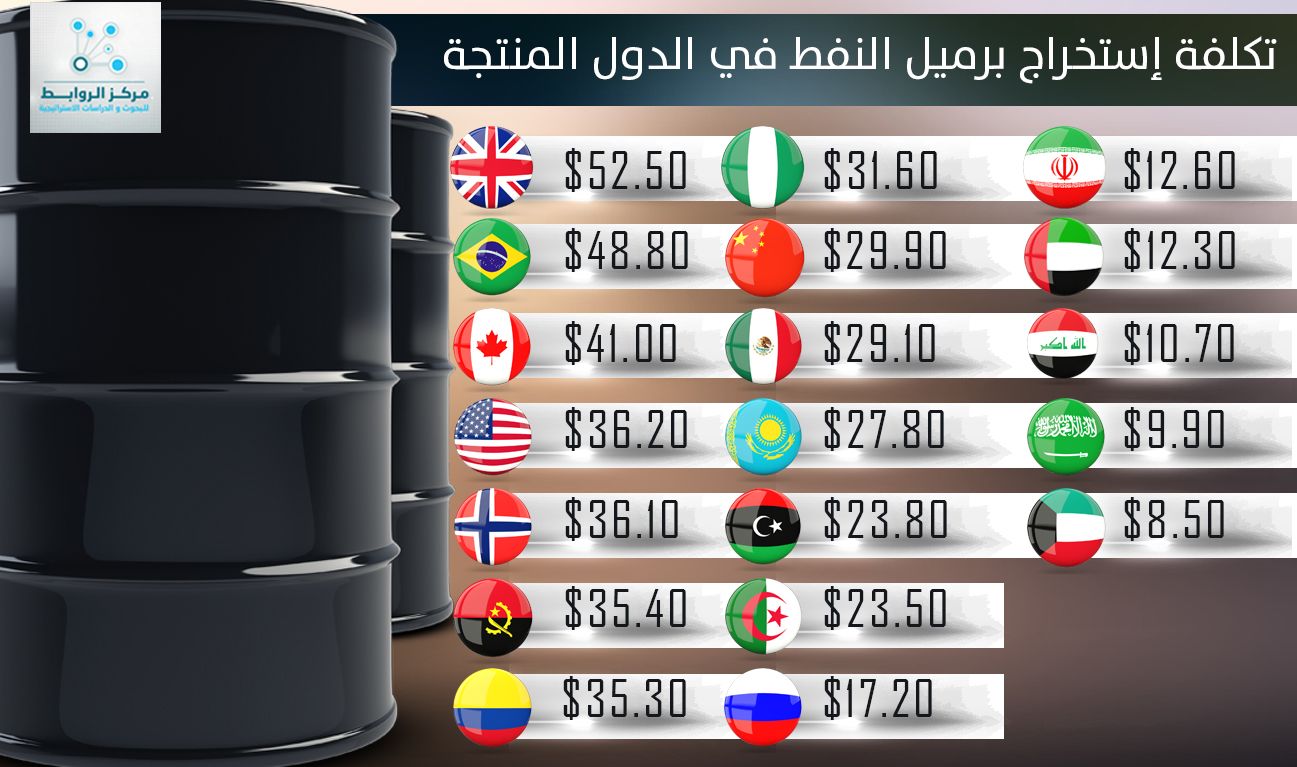
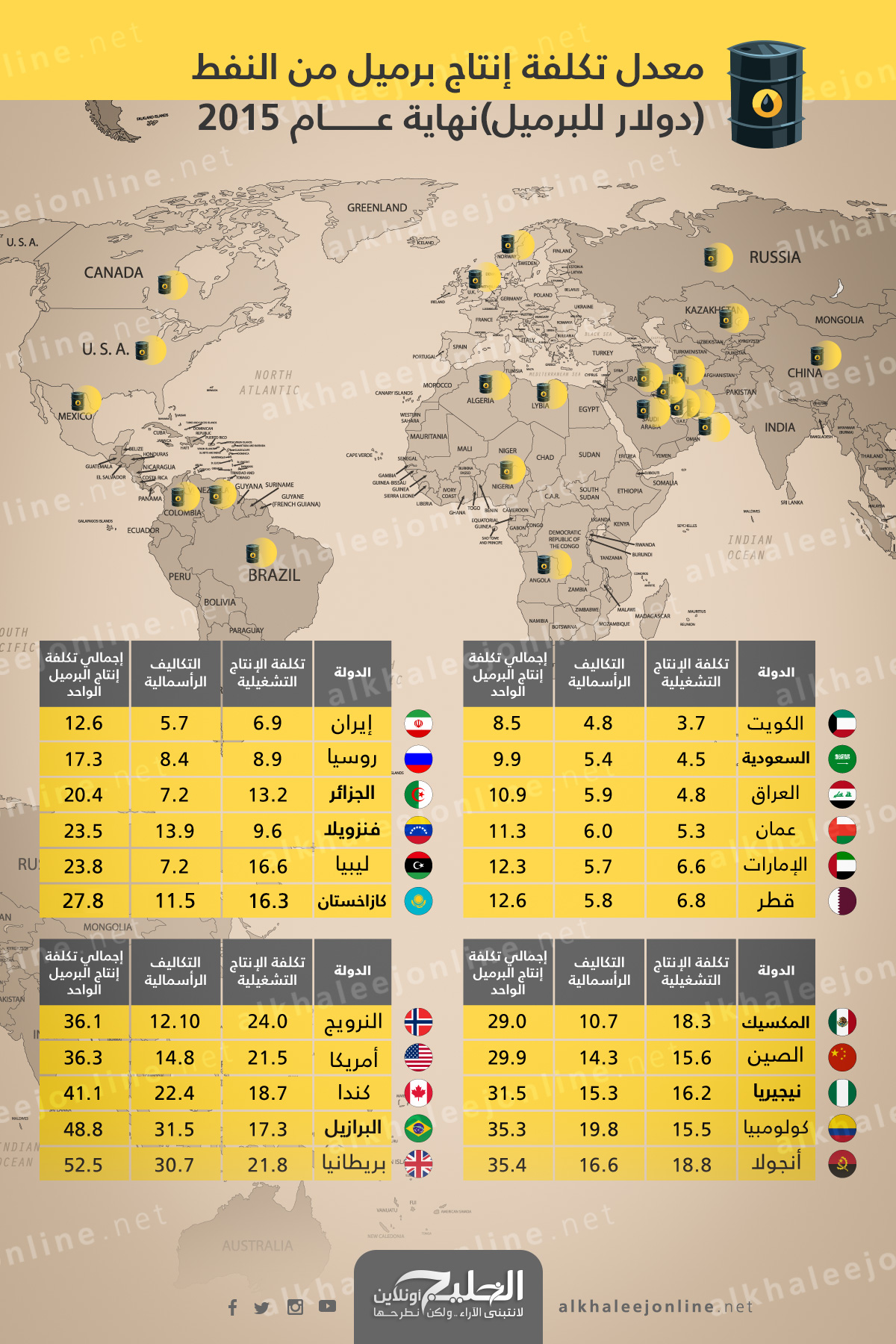
انفوجرافيك "الوسط"... تعرف على تكلفة إنتاج برميل النفط حول العالم؟ | اقتصاد - صحيفة الوسط البحرينية - مملكة البحرين
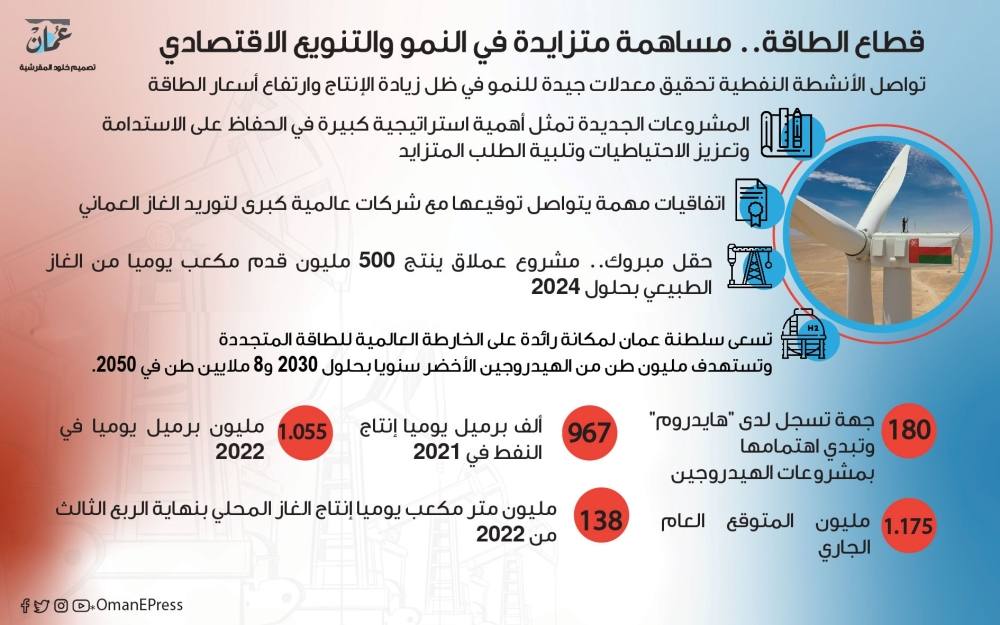
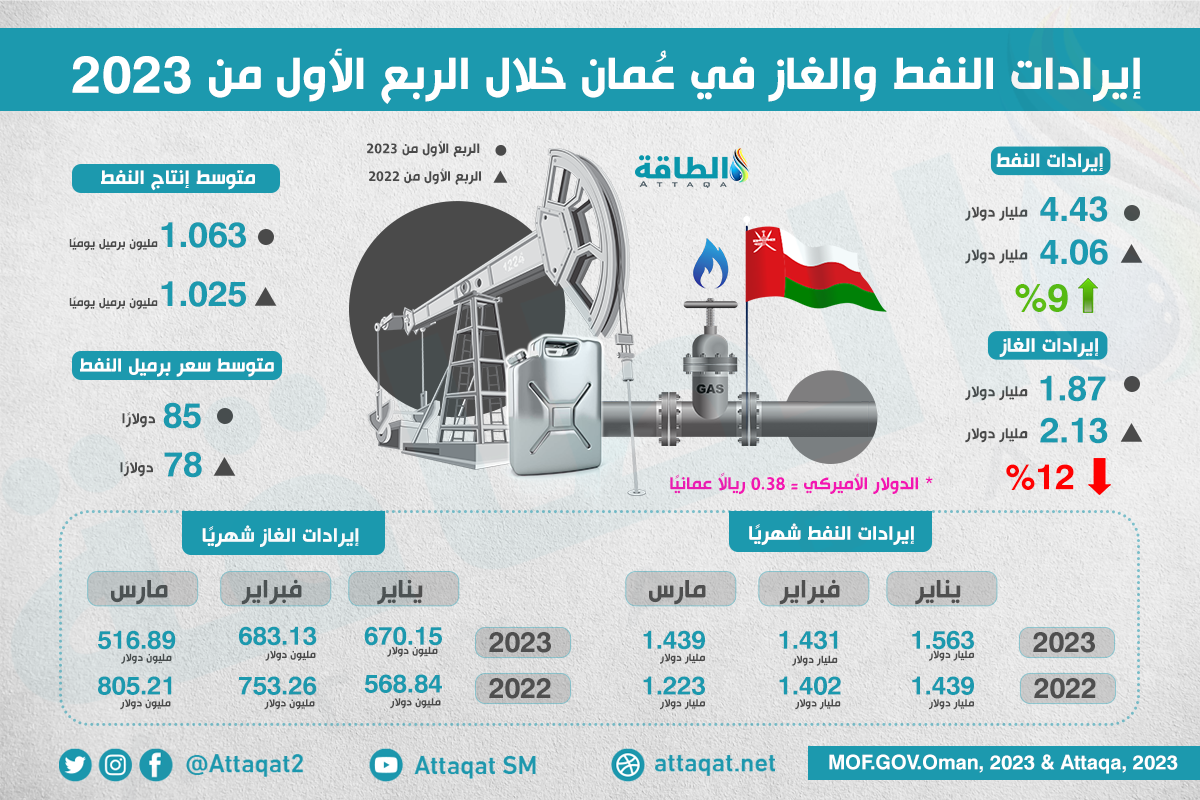
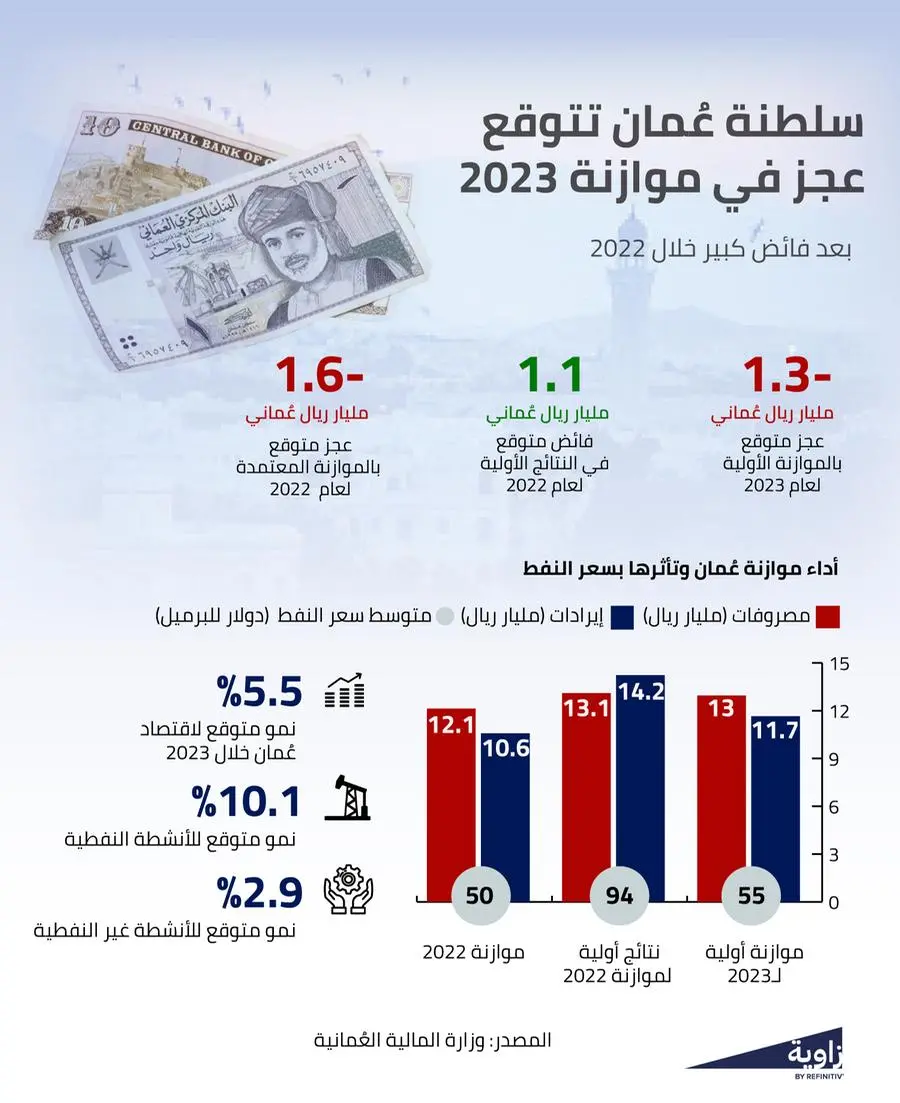
خام عمان يتجاوز 84 دولارا.. و 120 ألف برميل يوميا زيادة متوقعة في إنتاج 2023 - الموقع الرسمي لجريدة عمان

عالم النفط والغاز - انفوغرافيك يوضح معدل كلفة إنتاج برميل النفط في عدد من الدول حول العالم. #النفط #النفط_والغاز #معلومة_نفطية #إنتاج_النفط #اقتصاد | Facebook

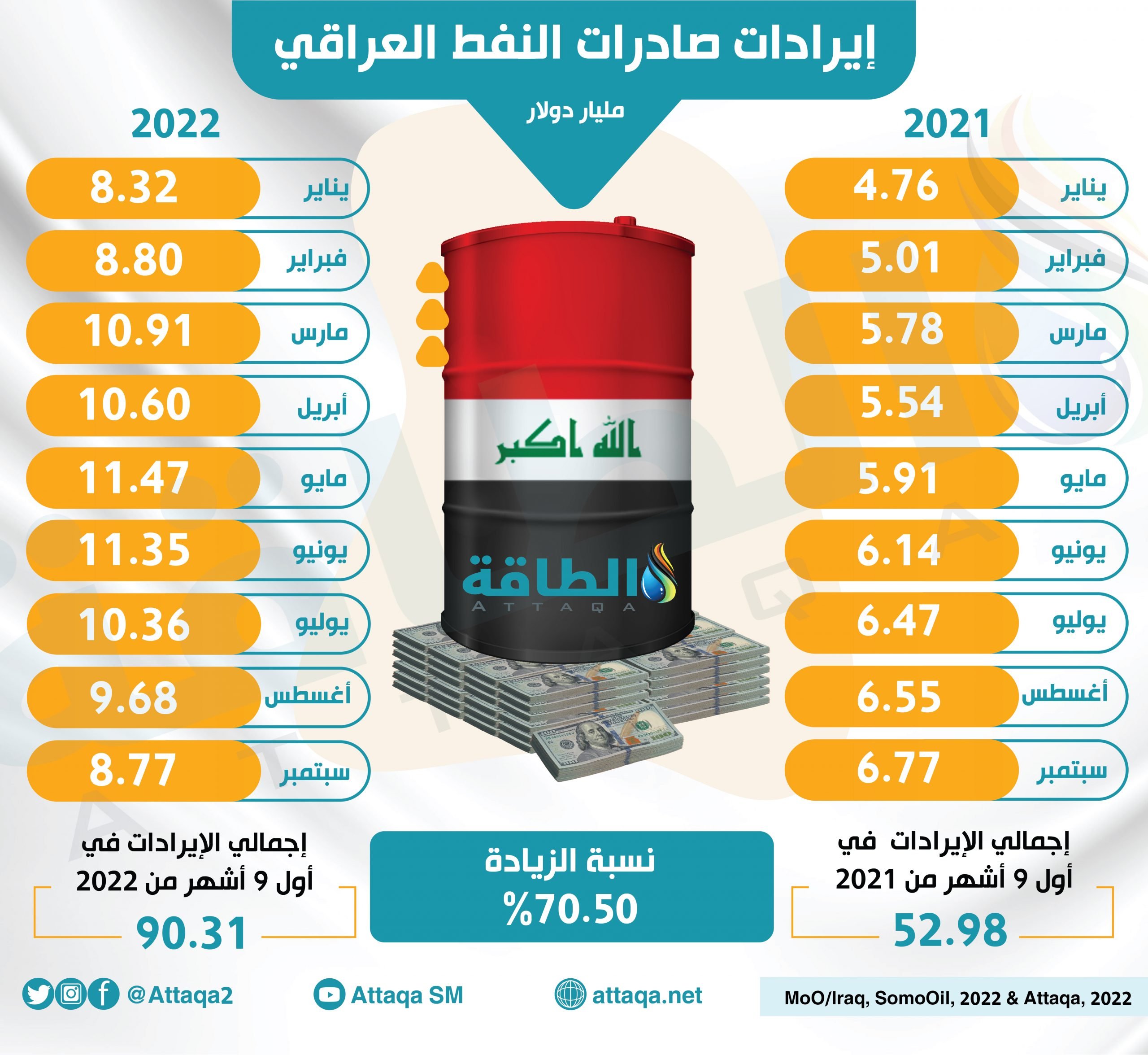
موقع بوابة الأخبار | بلغ إجمالي كمية صادرات #سلطنة_عُمان من #النفط حتى نهاية شهر يوليو 2023م نحو 178 مليونًا و956 ألفًا و300 برميل باحتساب متوسط سعر البرميل عند 80.5 دولار أمريكي، بينما
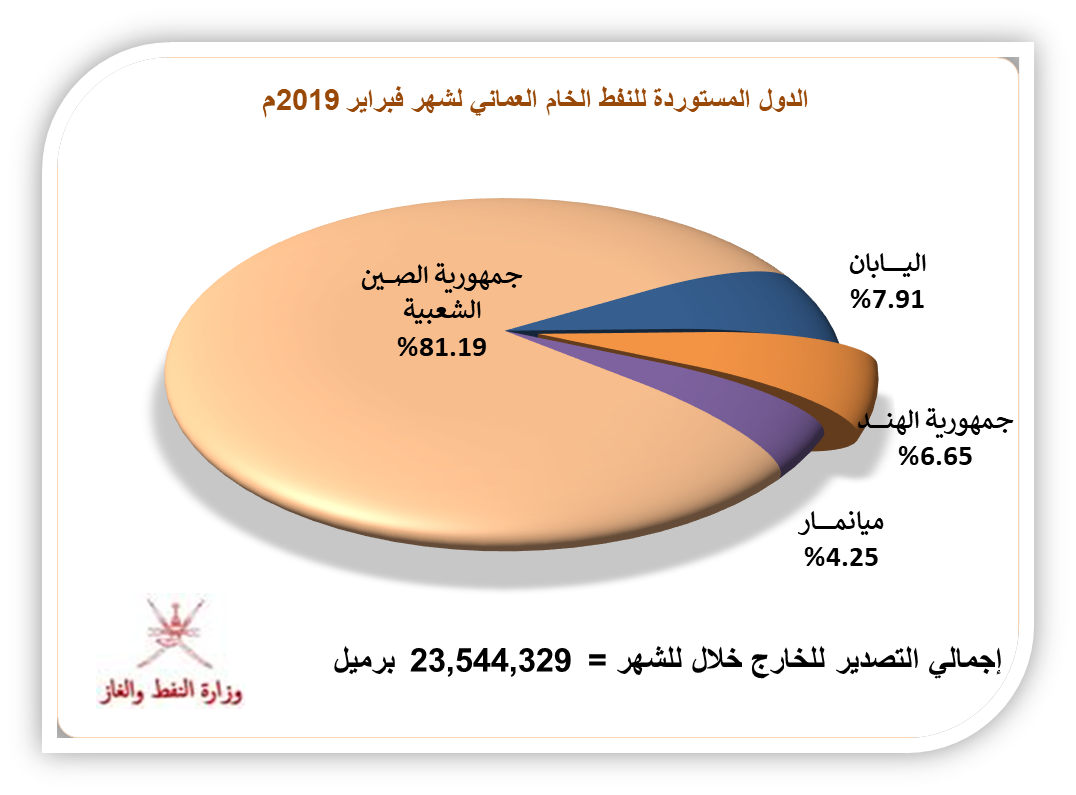
هذا هو حجم إنتاج السلطنة من النفط الخام والمكثفات النفطية في فبراير 2019 - الشبيبة | آخر أخبار سلطنة عمان المحلية وأخبار العالم




(88).jpeg)