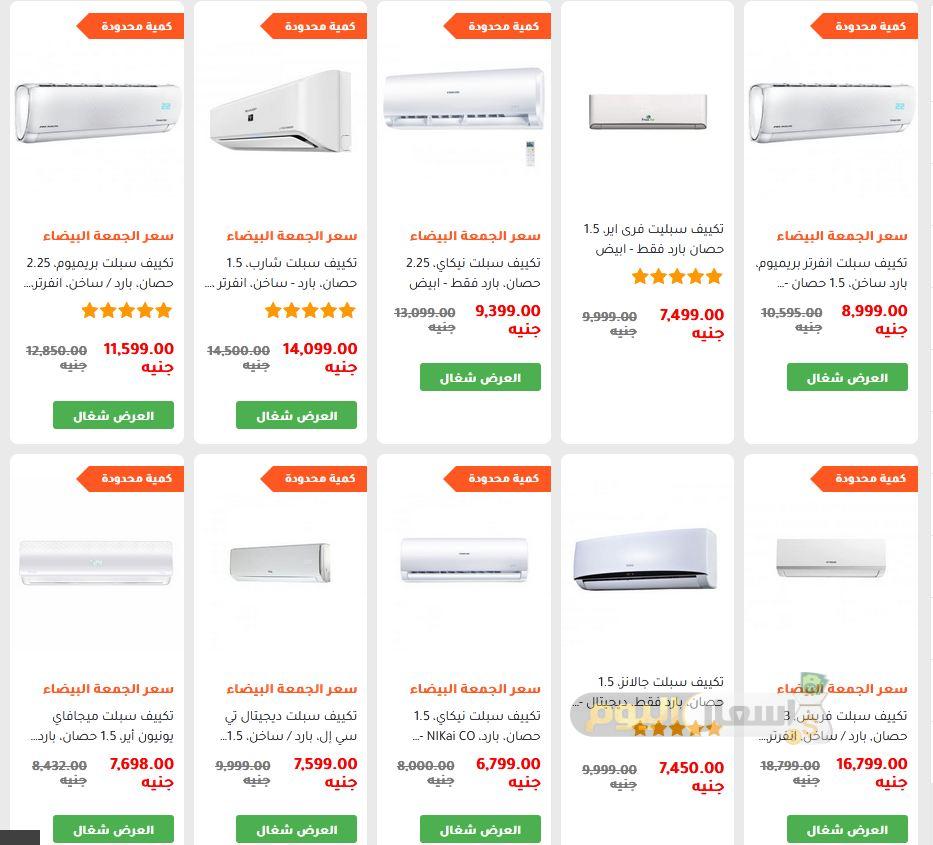
احصل على تكييف سبلت ميديا، 1.5 حصان، بارد/ساخن، انفرتر، Momb-12Hr-Dn - ابيض مع أفضل العروض | رنين.كوم

احصل على تكييف سبلت شارب، 2.25 حصان، بارد/ساخن، انفرتر، بلازما كلاستر، Y-XP18YHER - احمر مع أفضل العروض | رنين.كوم

احصل على تكييف سبلت كارير اوبتي ماكس، انفرتر، 3 حصان، بارد فقط، KHCT24DN-708F - ابيض مع أفضل العروض | رنين.كوم

احصل على تكييف سبلت انفرتر بلازما شارب، بارد، 1.5 حصان، ديجيتال، AH-AP12UHEA - ابيض مع أفضل العروض | رنين.كوم

الأجهزة الكهربائية في #رنين هتلاقيها باحسن سعر و جودة عالية و ماركات متنوعة💪🏻❤️ متوفر من الخميس الي السبت 8-9-10 ديسمبر فى جميع… | Instagram